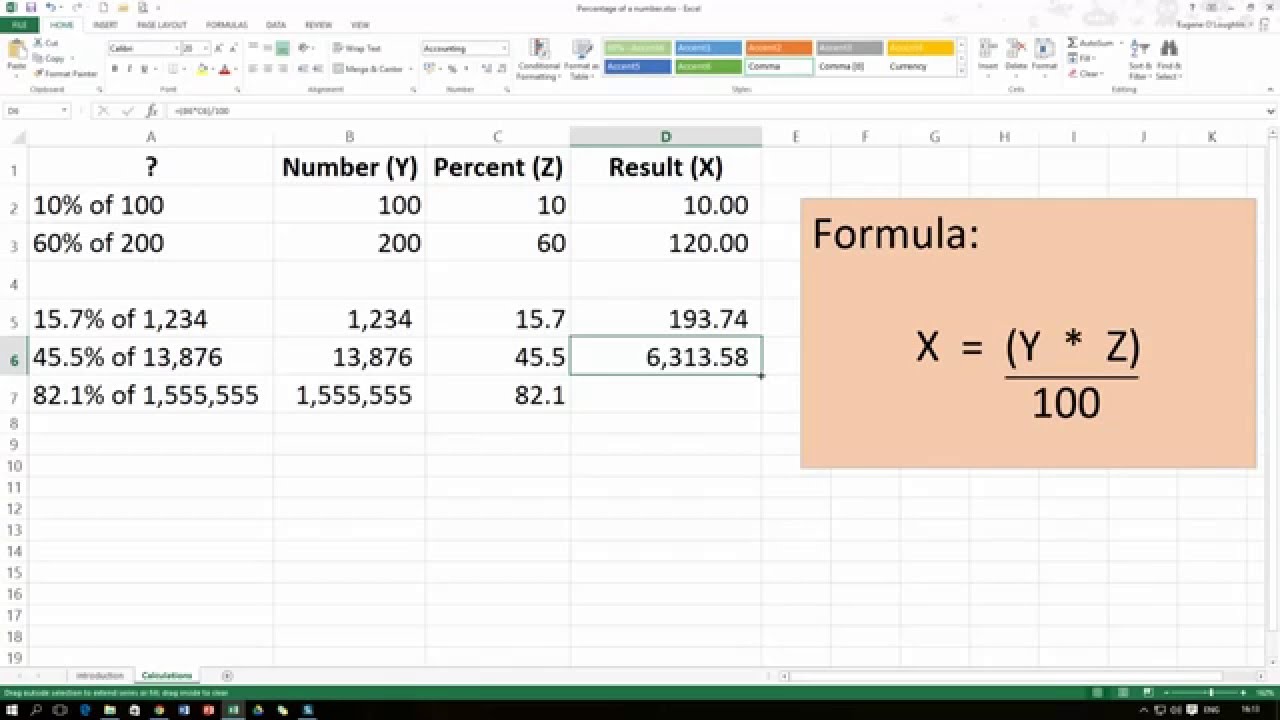 
It's easy to do this calculation on any scientific calculator because it will have a 'log' button. Or, the molar hydrogen ion concentration equals 10 to the power of the negative pH value. In other words, pH is the negative log of the molar hydrogen ion concentration. The equilibrium equation yields the following formula for pH: Using the concentration of hydrogen and hydroxide ions, the following relationship results:īasic Solution: < 1x10 -7 How to Calculate pH and If you are given concentration in any other unit (mass percent, molality, etc.), convert it to molarity in order to use the pH formula. Molarity is expressed in units of moles of solute per liter of solution (not solvent). When calculating pH, remember that refers to molarity, M. When water dissociates it yields a hydrogen ion and a hydroxide. There are several ways to define acids and bases, but pH only refers to hydrogen ion concentration and is only meaningful when applied to aqueous (water-based) solutions. To calculate it, take the log of the hydrogen ion concentration and reverse the sign to get the answer. This means pH is the negative base 10 logarithm ('log' on a calculator) of the hydrogen ion concentration of a solution. A value less than 7 is acidic, while a value greater than 7 is basic. pH is a measure of how acidic or basic a chemical solution is.Here's a quick review of how to calculate pH and what pH means with respect to hydrogen ion concentration, acids, and bases.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
